Abstract
Introduction
Venetoclax in combination with a hypomethylating agent (VEN-HMA) has become a standard of care for older or unfit patients with newly diagnosed AML. Although primarily administered in the outpatient setting, VEN-HMA is associated with significant cytopenias and infectious complications, requiring careful monitoring and dose adjustments. Patients treated with VEN-HMA on clinical trials had a median age of ~75 years. The optimal dose and schedule, safety, and efficacy of VEN-HMA in octo- and nonagenarians is not clearly defined.
Methods
We performed a retrospective analysis of AML patients ≥80-years-old who received at least 1 day of VEN-HMA at our institution from 11/2018 to 7/2021. Patients with de novo or secondary AML with or without prior HMA or chemotherapy were included. Venetoclax starting dose was 200-400 mg for 14-28 days. Dose adjustments for drug interactions with azole anti-fungals were implemented. HMA was started at 50-75 mg/m2 for 5-7 days of azacitidine or 20mg/m2 for 5 days of decitabine. Dose reduction was defined as any decrease from the starting dose and schedule. Patients who did not complete cycle 1 were included in the overall survival and safety analysis but excluded from response assessment.
Results
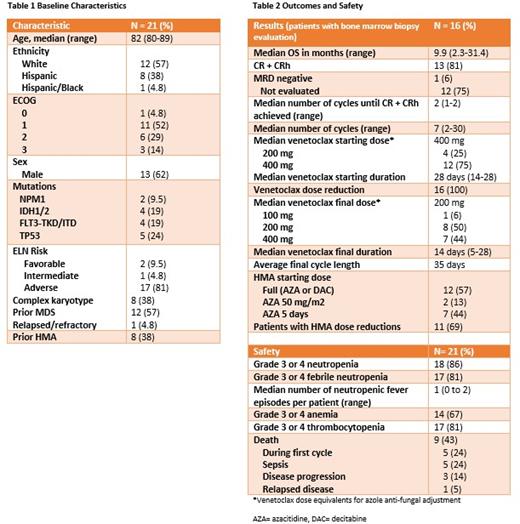
Among 21 patients ≥80-years-old treated with VEN-HMA (20 newly diagnosed, 1 relapsed/refractory), median age was 82 years (range: 80-89) (Table 1), 57% had antecedent MDS, and 38% received HMAs previously. Most patients (81%) were ELN adverse risk, 38% had a complex karyotype, 24% had a TP53 mutation, and 43% had ECOG PS of 2-3. Median overall survival for all patients was 8.0 months (0.5-31.4 months). At time of analysis, 12 patients (57%) were still alive and in remission on VEN-HMA with a median follow-up of 11.5 months (range 2.3-31.4 months). Five patients (24%) died during cycle 1 from sepsis. Of these 5 patients, 4 had a TP53 mutation, 3 had prior MDS, and 3 had received prior therapy for lymphoma. In the remaining 16 patients, median overall survival was 9.9 months (2.3-31.4 months) and the CR/CRh rate was 81% (13/16 patients). Median duration of response was 8.9 months (range 1.0-30.0). Consistent with previous reports, all patients who achieved CR/CRh did so by the end of cycle 2 (median 2 cycles). Most patients also received the standard dose and schedule of VEN (75%) and HMA (57%) during the first cycle. All patients (100%) required venetoclax dose and schedule reduction, with a median final venetoclax dose of 200 mg and duration of 14 days. Average final cycle length was 35 days. Most patients (69%) also required dose reduction of HMA. Median duration of treatment was 7.5 months (range 0.5-31.4). Treatment emergent grade 3-4 anemia occurred in 67% of patients, thrombocytopenia in 81%, and neutropenia in 86%; 17 patients (81%) had treatment emergent febrile neutropenia. There were no infectious deaths in patients who survived cycle 1. The median number of neutropenic fever episodes per patient was 1 (0 to 2). The 4 deaths after cycle 1 were due to progressive disease (n=3) or relapse (n=1).
Conclusion
VEN-HMA can be safely and effectively given to octogenarians with careful monitoring and dose adjustments. All patients required dose reduction of venetoclax after CR/CRh was achieved, and most also required adjustment of HMA. Despite this, over half of patients achieved durable remissions. The greatest risk of infectious death was in the first cycle, in patients who were heavily pre-treated and enriched for TP53 mutations. When compared to historical controls, outcomes in octogenarians who survive the first cycle appear similar to younger age groups. This work highlights the need for a prospective multi-center effort to optimize the dose and schedule of VEN-HMA in older patient populations.
Bradley: AbbVie: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees. Sekeres: Takeda/Millenium: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees. Watts: Rafael Pharmaceuticals: Consultancy; Genentech: Consultancy; Bristol Myers Squibb: Consultancy; Takeda: Consultancy, Research Funding; Jazz Pharmaceuticals: Consultancy; Aptevo Therapeutices: Research Funding.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal